
SciELO - Brasil - Modification of Chitosan by Zincke Reaction: Synthesis of a Novel Polycationic Chitosan-Pyridinium Derivative Modification of Chitosan by Zincke Reaction: Synthesis of a Novel Polycationic Chitosan- Pyridinium Derivative

Pyridinium Salts as Redox‐Active Functional Group Transfer Reagents - Rössler - 2020 - Angewandte Chemie International Edition - Wiley Online Library

Pyridinium Salts as Redox‐Active Functional Group Transfer Reagents - Rössler - 2020 - Angewandte Chemie International Edition - Wiley Online Library
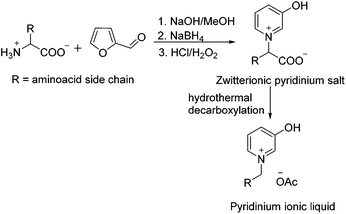
Pyridinium salts: from synthesis to reactivity and applications - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C7QO00836H
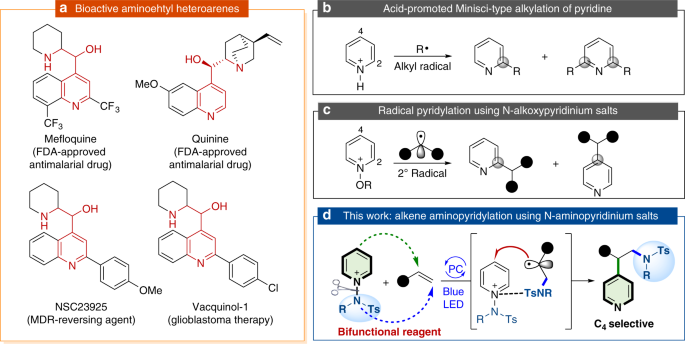
Visible light induced alkene aminopyridylation using N-aminopyridinium salts as bifunctional reagents | Nature Communications
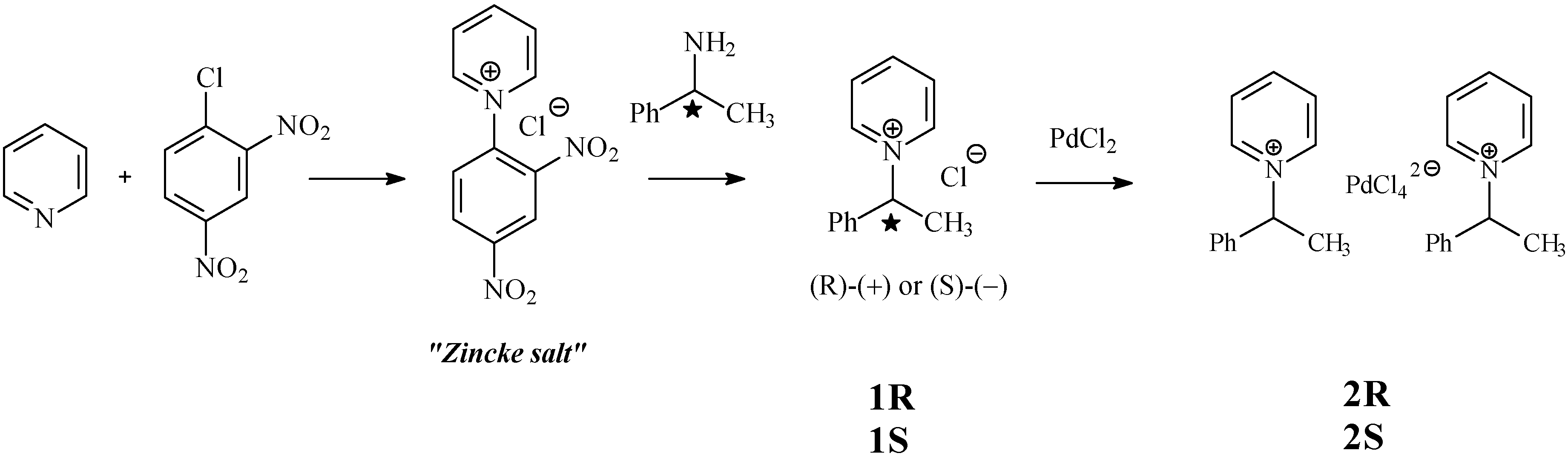
Molecules | Free Full-Text | Palladate Salts from Chiral Pyridinium Ionic Liquids: Synthesis and Crystal Structures
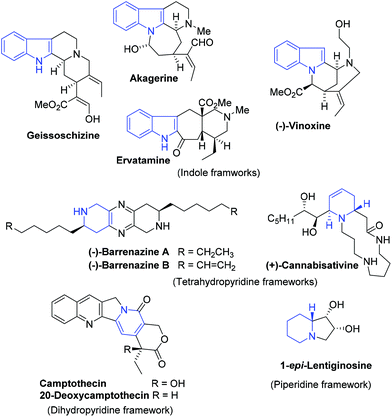
Pyridinium salts: from synthesis to reactivity and applications - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C7QO00836H

Synthesis of chiral piperidines from pyridinium salts via rhodium-catalysed transfer hydrogenation | Nature Catalysis

J. Am. Chem. Soc. on Twitter: "Donor–Acceptor Pyridinium Salts for Photo-Induced Electron-Transfer-Driven Modification of Tryptophan in Peptides, Proteins, and Proteomes Using Visible Light @UWArtsSciences @Novartis @NovartisScience @WYSpaceGrant ...
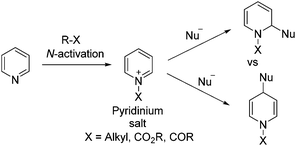
Pyridinium salts: from synthesis to reactivity and applications - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C7QO00836H

Pyridinium salts: from synthesis to reactivity and applications - Organic Chemistry Frontiers (RSC Publishing)













